Overview
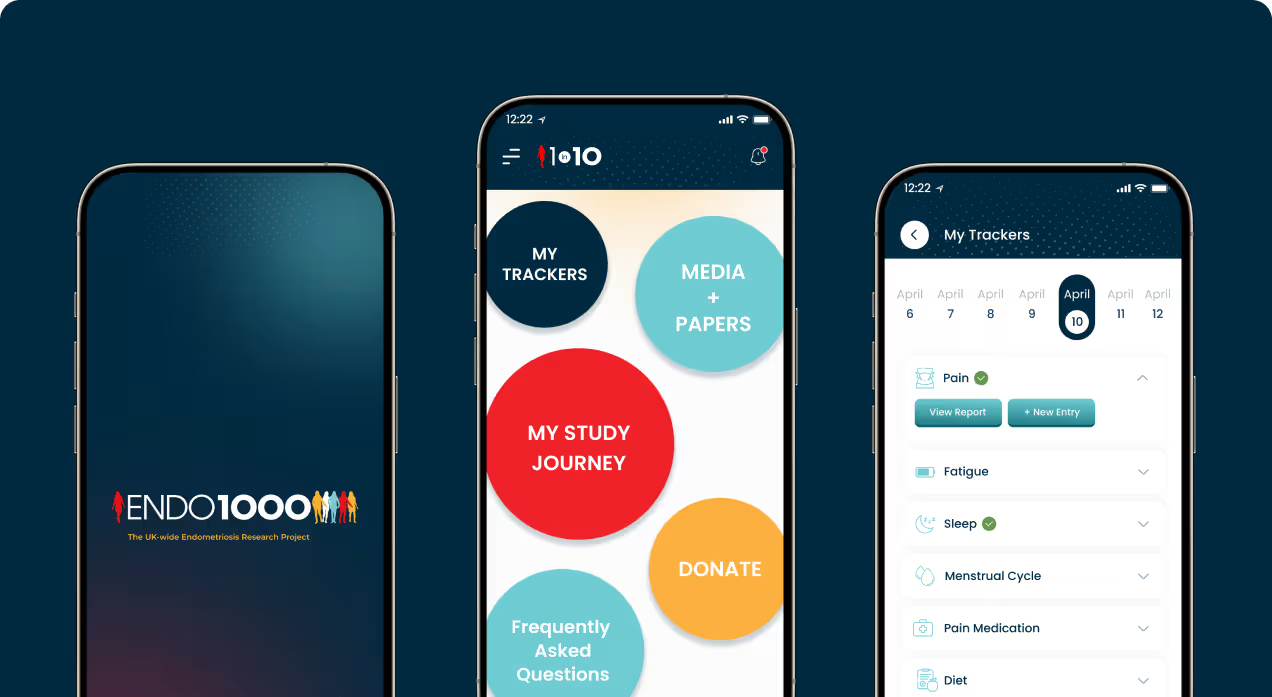
ENDO1000 was developed as part of a research collaboration led by academic and clinical researchers to collect structured health data from approximately 1,000 participants. Intelivita developed a mobile application, supporting website, and web-based administration platform that enables participants to submit symptom data through guided questionnaires, track health metrics, and access research information while securely transmitting data to university research infrastructure.

The Challenge
The project involved 5 primary challenges..
Long-Term Data Collection
Participants needed to submit consistent symptom data over extended research periods.
Research Participation
The platform required mechanisms to keep participants engaged in ongoing studies.
Multi-Study Management
Researchers needed tools to manage multiple studies and participant groups.
Clinical Data Quality
Symptom submissions required structured input to maintain research-grade standards.
Patient Insight Visibility
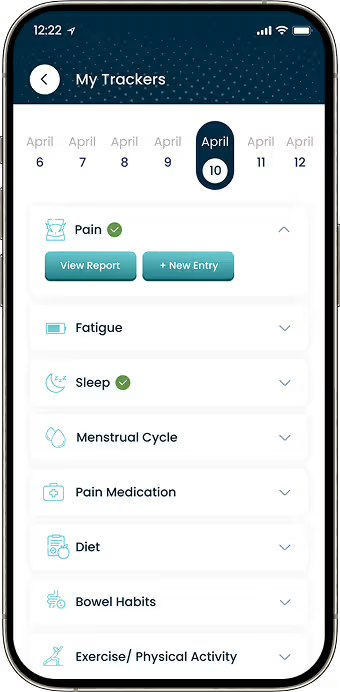
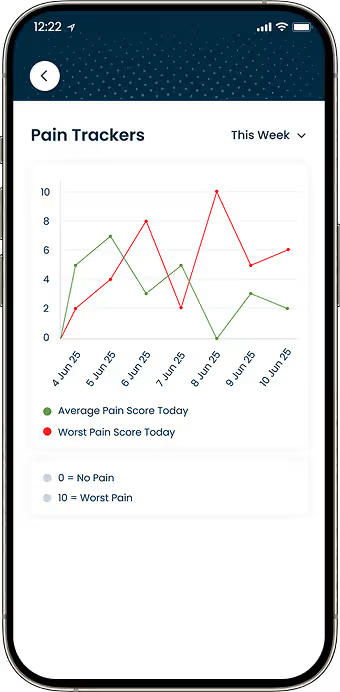
Participants needed visual reports to understand changes in their condition over time.
The Solutions
To overcome the challenges, five key solutions were developed and executed with precision.
Structured Symptom Tracking
Developed guided questionnaires to capture consistent symptom data.
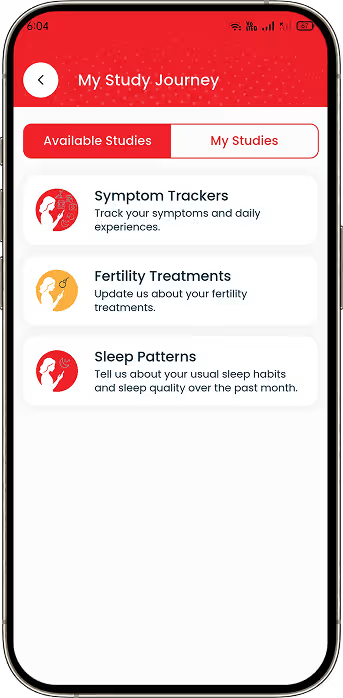
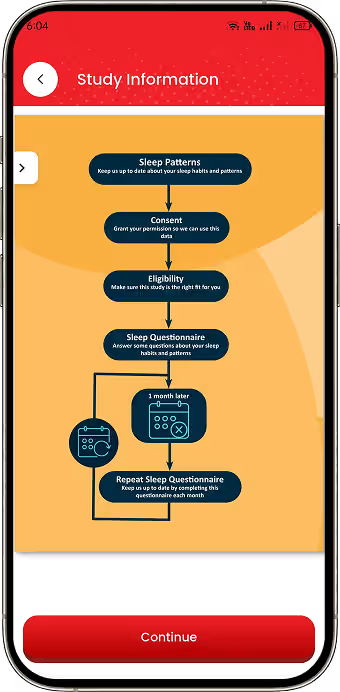
Digital Study Enrolment
Implemented eligibility checks and digital consent workflows for research participation.
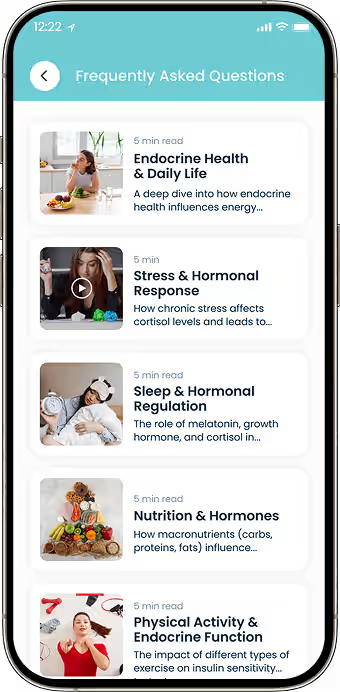
Health Data Visualization
Built dashboards that display symptom trends across time periods.
Centralized Administration
Delivered an admin system for managing studies, participants, and research content.
Engagement Workflows
Introduced reminders and simplified daily logging to support long-term participation.
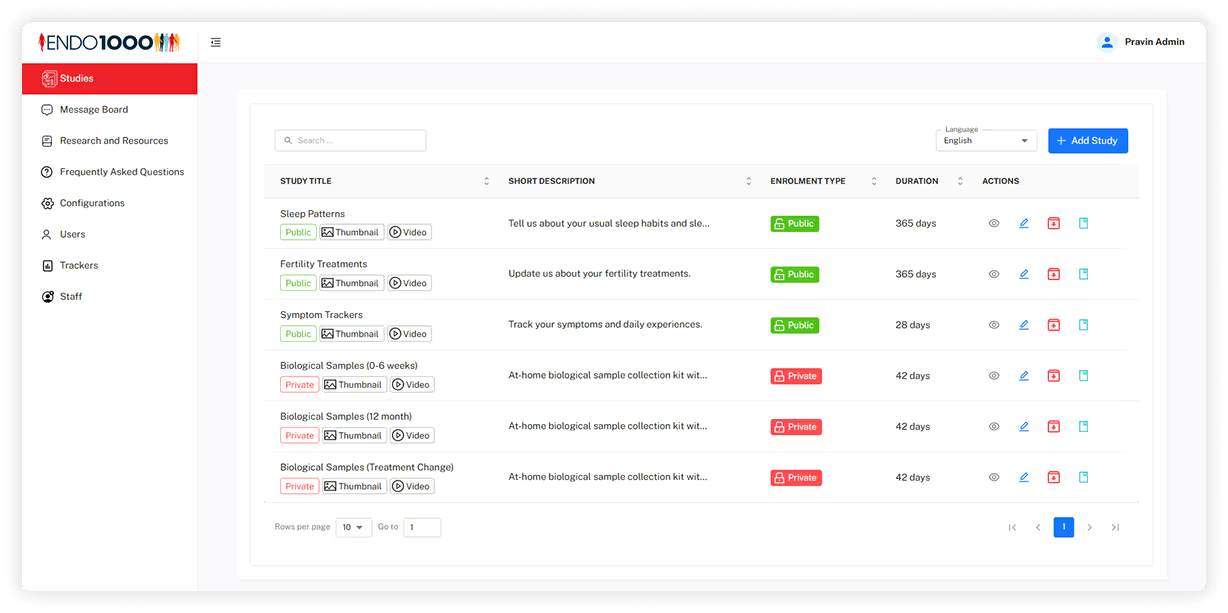
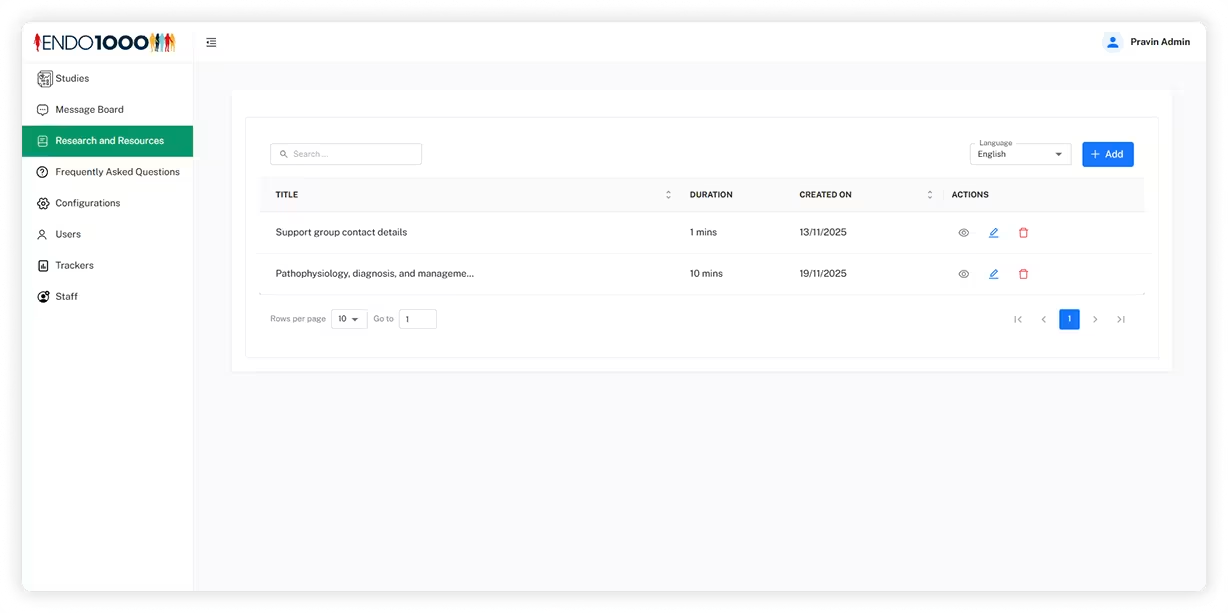
Admin Panel
Intelivita developed a web-based administration system that enables research teams to manage studies, participants, symptom trackers, content resources, announcements, and staff permissions while monitoring participant activity across research programmes.
Technology Stack
The Result
Intelivita delivered ENDO1000 as a digital research platform that enables participants to submit structured endometriosis symptom data remotely. The system supports consistent patient data collection, provides visual symptom tracking, and enables researchers to manage clinical studies through a centralized administration platform.